Twenty-five years ago, William E. Moerner was investigating the potential for biological data storage at IBM. During his research, he became the first person to image a single molecule.
On Wednesday, Moerner was awarded the Nobel Prize in Chemistry for work contributing to the development of super-resolution microscopy – work that all started with that discovery at the IBM lab.
Unfortunately Moerner (known as W.E.) wasn’t home to receive the call from Sweden, instead learning the news from his wife while at a conference in Brazil.
“Of course, your heart races and you say ‘Can this be? Can this be?’” he said to nobelprize.org from Brazil. “I’m incredibly fortunate.”
The $1.1 million prize is shared between Moerner, Eric Betzig of Virginia’s Howard Hughes Medical Institute and Stefan Hell of Germany’s Max Planck Institute. Moerner is the Harry S. Mosher Professor in Chemistry and is a professor, by courtesy, of applied physics at Stanford.
“He’s always excited,” said Gabriela Schlau-Cohen, a former Moerner lab post-doctoral scholar who recently left to become an assistant professor at MIT. “He’s been in this business a long time, but he’s still always excited about new results. If you’ve been struggling to get an experiment to work and you finally have signal, he loves it if you knock on his door or send him an email.”
Moerner and Betzig were awarded for work done separately to advance super-resolution microscopy, while Hell was awarded for developing a technique that uses laser beams to achieve similar high-resolution imaging results.
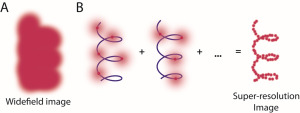
The research into super-resolution microscopy has enabled researchers to effectively image things that are smaller than a wavelength of light. That size had been accepted as the limit of microscopy’s ability because optical microscopes by definition cannot resolve objects smaller than that.
To perform super-resolution microscopy, a researcher first must label the molecules of interest with something fluorescent. However, for this labeling to be useful the fluorescent molecule must be switchable – that is, the researcher must have a mechanism by which to turn the light on and off. While at UC-San Diego in the 90s, Moerner discovered just such a molecule.
Previous fluorescent molecules used in research could be turned on with a specific wavelength of light, but they could only be turned on once. The variant of green fluorescent protein (GFP) that Moerner discovered could be turned on by one wavelength of light and, after fading, reactivated by another.
Once the molecule of interest is labeled, researchers provide a sequence of small light stimuli while capturing images of the cell. Not every GFP tag will illuminate at the same time, so researchers can distinguish individual molecules labeled with GFP as spots of light begin to appear and then fade after stimulation.
While these spots are larger than the molecules themselves, an approximation of the spot’s center provides the location of the labeled molecule. To build a full image, simply superimpose all of the locations of center onto one image. The uncertainty present in approximating the spot’s center is less than the uncertainty of blurry optical microscope images, resulting in a potentially tenfold increase in resolution.
One key application of super-resolution microscopy is the ability it allows biologists to observe the different activities in various bacterial compartments, since optical microscopy can only produce blurred images of bacteria due to their size.
The Moerner lab is collaborating with Professor Lucy Shapiro’s lab in the medical school to investigate bacterial processes. The team is looking at how Caulobacter crescentus divide to gain further insight into important aspects of bacterial pathogenicity, including bacterial multiplication, division and colonization.
The lab is also using super-resolution microscopy to investigate cilia on cell surfaces, which are about as long as one wavelength of light (the limit of optical microscopy resolution).
Another biological application is the ability to observe different parts of processes that occur asynchronously in nature and that cannot be synchronized in the lab. By tracking a molecule of interest, researchers can observe each intermediate in the process and thus learn more about the process as a whole.
Schlau-Cohen, who recently left the Moerner lab for MIT, researches the early events in photosynthesis such as how photosynthetic systems absorb energy via photons and convert that energy to electricity. The ability to resolve images to the molecular level is key because it allows her to observe the heterogeneity of individual proteins participating in the photosynthetic process process.
“One of the things that’s really amazing about W.E. is he has this amazing experimental intuition for thinking about how an experiment works, what your signal could be coming from,” she said. “He has an incredible technical expertise, and I think that it’s that – not just skill, but talent – that allowed him to detect the first molecule.”
Yoav Shechtman, a post-doc in the Moerner lab, is working on the lab’s project to expand 2D super-resolution microscopy into 3D. He said that Moerner is an excellent mentor in part because he can balance the big picture with important details.
“Somehow he manages to know exactly what everybody is doing and always has very good insight and very good experimental intuition,” Shechtman said. “He has the nose to smell what’s important and what’s the direction to go to.”
Graduate student lab member Marissa Lee was inspired to pursue physical chemistry instead of organic chemistry after working as one of Moerner’s teaching assistant’s for Chemistry 31X, an accelerated introductory chemistry course.
“He was such a good teacher, and everybody in the lab seemed to be very supported and have a good community, so I ended up joining this lab and doing this research for W.E,” Lee said. “I think you should always join a lab for the adviser.”
Matt Lew M.S. ’10 Ph.D. ’14 did just that, sending Moerner an unsolicited e-mail that opened the door for Lew, an electrical engineering graduate student at the time, into the lab. Moerner would become Lew’s dissertation adviser and recommend Lew for a faculty position at Washington University in St. Louis, where Lew will start as an assistant professor next year.
Moerner earned two B.S. degrees (in Physics and Electrical Engineering) and a B.A. (in Mathematics) at Washington University in St. Louis, before completing his graduate work at Cornell. The Pleasanton, California, native started at Stanford in 1998.
He becomes the fifth Stanford faculty member to win the Nobel in the past three years, joining Michael Levitt (2013 – Chemistry), Thomas Südhof (2013 – Medicine), Alvin Roth M.S. ’73 Ph.D ‘74 (2012 – Economic Sciences) and Brian Kobilka (2012 – Chemistry).
This post has been updated. Professor Moerner’s courtesy appointment was previously misidentified as applied sciences. He is a professor, by courtesy, of applied physics. The Daily regrets this error.
